About FQAD
Definition and diagnostics
Fluoroquinolone-Associated Disability – also known as FQAD – is a disease with severe, long-lasting and potentially irreversible side effects caused by treatment with a fluoroquinolone antibiotic. The diagnostic criteria of FQAD, chosen by the FDA (1), are:
- Substantial limitation to perform normal life functions.
- Two or more of the following systems are affected:
- Musculoskeletal system
- Peripheral nervous system
- Skin
- Cardiovascular system
- Sensory organs
- Neuropsychiatric
- The side effects persist for more than 30 days after discontinuation of the medication.
Official recognition of FQAD
Unfortunately, there is currently no diagnosis code from the WHO, which means that there is no suitable ICD code. This is one of the concerns that the Association for the Clarification of the Side Effects of Fluoroquinolone Antibiotics (VFCN) has integrated into its mission and objectives (2). Without an ICD code, it is difficult for those affected to gain recognition from doctors and insurance companies. Another attempt is currently underway to obtain at least a subcode in the ICD, as the code T36 Poisoning by systemic antibiotics already exists, with several subcodes such as T36.0 Poisoning: penicillins. (13) Here, an extension as T36.7 Poisoning: Fluoroquinolones would be desirable.
Official warning from the EMA regarding fluoroquinolones
In 2019, the European Medicines Agency, or EMA for short, held a public consultation in Europe on fluoroquinolones, with the decision to issue a new warning letter to doctors, a so-called red hand letter, as well as further indication restrictions (3). Here is a small excerpt:
“08.04.2019 Systemically and inhaled quinolone and fluoroquinolone antibiotics: Risk of long-lasting and potentially irreversible side effects affecting quality of life – Restrictions on use.
Long-lasting and possibly irreversible side effects affecting the quality of life have been reported in connection with antibiotics containing quinolones and fluoroquinolones. They mainly affect the musculoskeletal system and the nervous system.”
Pathomechanism and symptoms
There is now more literature on the pathomechanism and potential treatment options for this disease. The essential “Fluoroquinolone-Associated Disability FQAD: Pathogenesis, Diagnostics, Therapy and Diagnostic Criteria” (4) by Dr. Stefan Pieper, MD, published in 2020, provides a good overview of the current state of research.
According to Dr. Stefan Pieper, FQAD affects the following four body systems, which can be affected to varying degrees depending on the patient:
- Mitochondrial dysfunction due to increased oxidative stress and inhibition of topoisomerase II.
- Collagen damage due to upregulation of various matrix metalloproteinases
- Damage to the peripheral nervous system due to oxidative stress, mitochondrial dysfunction or direct neurotoxic effects.
- Neuropsychiatric side effects due to fluoroquinolone-induced inhibition of the GABAergic system and excessive activation of NMDA receptors.
This results in a wide-ranging clinical picture with many possible symptoms:
- Severe fatigue and exhaustion
- Tendinopathy, tendon rupture
- Increased risk of aortic aneurysms
- Damage to peripheral nerves and resulting paraesthesia, dysaesthesia and nerve pain
- Muscle weakness, myalgia, fasciculations
- arthralgia
- Sleep disorders, depersonalization and even suicidal tendencies




Potential therapy options
The excellently written paper by Krzysztof Michalak, PhD “Treatment of FQAD: Pathobiochemical Implications” (5) should also be mentioned here. In it, Krzysztof Michalak, PhD postulates the following potential treatment options:
- Reduction of oxidative stress.
- Restoration of the reduced mitochondrial potential.
- Supplementation of monovalent and divalent cations, which are chelated by Fluoroquinolones and probably transported ineffectively to the cell.
- Stimulation of mitochondrial proliferation.
- Removal of permanently accumulated Fluoroquinolones from the cell
- Regulation of disturbed gene expression and enzyme activity.
Nevertheless, Krzysztof Michalak PhD emphasizes that the molecular activity of fluoroquinolone antibiotics in the cells is largely unclear in many details. Unfortunately, this is still the case today, as little research has been carried out on FQAD compared to other diseases.
Dysfunction of GABAergic neurotransmission in FQAD
Another starting point for the treatment of FQAD is the antagonistic effect of Fluoroquinolones on the GABA-A receptor described by Dr. Stefan Pieper (4) and reported in various studies, which has been described as particularly enhanced when administered simultaneously with an NSAID (6). In a recent study, the effect on the GABAergic nervous system was also described in connection with chronic dysfunction of gastric and intestinal peristalsis. The reduced GABAergic transmission from the brainstem to the dorsal motor vagus nerve nucleus, which is crucial for the regulation of muscles, in this case the stomach and intestines, is the cause of the sometimes long-term symptoms of the gastrointestinal tract (12). From previous observations of patients treated by their doctors with antiepileptic drugs such as gabapentin, pregabalin or clonazepam, it has often been possible to significantly reduce quality of life-limiting side effects such as pain, paraesthesia, dysaesthesia and sleep disturbance, particularly at the beginning. Unfortunately, however, this has not yet been investigated in a study and is based solely on feedback from those affected. Of course, as with any other therapy, not all sufferers benefit from treatment with the above medications, and the side effects of long-term treatment with them should not be neglected. In any case, those antiepileptic drugs seem to be able to reduce the excitotoxicity (7), which is hypothetically triggered acutely by the antagonism of the Fluoroquinolone antibiotic at the GABA-A receptor. This seems to alleviate symptoms such as sleep disturbances, but also nerve pain. Another possibility appears to be an attempt to inhibit NMDA activity, as also described by Dr. med. Stefan Pieper (4). This could be attempted, for example, with the NMDA antagonist Memantine (8), or to a lesser extent with the non-competitive NMDA antagonist magnesium (9). Higher doses of magnesium in particular (from 600mg elemental magnesium) seem to help many people. Whether this effect is due to the effect on the NMDA receptor is unclear.


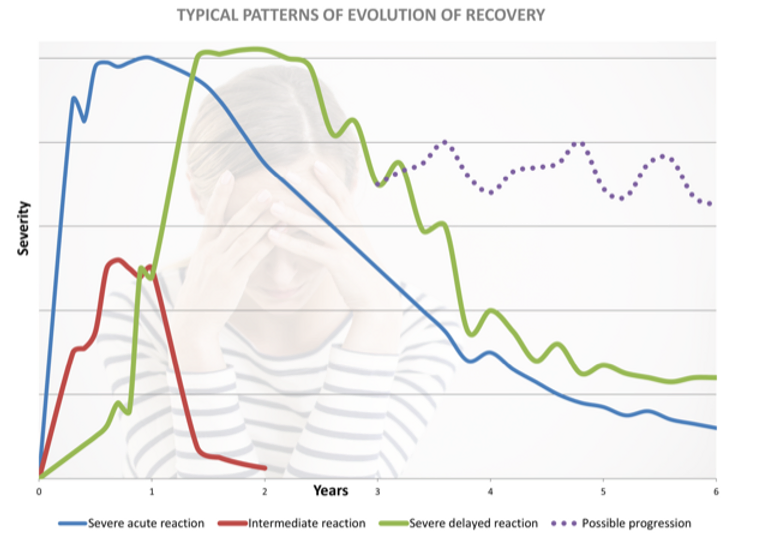
Progression of the Disease
The website of the Association for the Clarification of the Side Effects of Fluoroquinolone Antibiotics (VFCN) provides a good overview of the different courses of the disease: (10)
The severity and duration of the side effects vary from case to case. For example, some patients with severe physical limitations make a full recovery after a while, while others with initially only moderate side effects experience a progressive course of the disease. Based on the reported cases, the US Food and Drug Administration (FDA) assumes an average duration of 14 months for severe side effects. However, a number of cases of permanent damage have also been reported. The Flox report provides an overview of possible courses. It illustrates possible moderate to severe courses using three different curves (see below). The data is based on the observation of the course of the disease in 49 patients. Excluded from this study are mild and common side effects, which usually disappear after discontinuation of the medication. It is important to emphasize that the above-mentioned report was written by patients themselves and therefore cannot be considered to be of a high scientific standard. (11) Nevertheless, it offers an interesting overview of possible progressions, which has been confirmed in practice with FQAD patients. However, the report is already 14 years old. Many of the patients knew less about the causes and potential treatment options for FQAD, as there were hardly any studies on FQAD at that time.
Conclusion
In conclusion, despite some interesting studies and theories on FQAD, a great deal of knowledge is still needed to establish a precise cause and therapy. The question of which population with which individual conditions suffers from the severe, sometimes irreversible side effects is also unresolved. More research is therefore essential in order to offer help to those affected.
Marco Karrer M.D.
Updated October 2024
Sources:
M.A. Green, R.F. Halliwell Selective antagonism of the GABA A receptor by ciprofloxacin and biphenylacetic acid; 2009
GABA A Receptor Activation Attenuates Excitotoxicity but Exacerbates Oxygen-Glucose Deprivation-Induced Neuronal Injury In Vitro; 1996
Melinda K. Kutzing, Vincent Luo, Bonnie L. Firestein; Protection from glutamate-induced excitotoxicity by memantine
The International Journal of Neuropsychopharmacology; Bartłomiej Pochwat, Bernadeta Szewczyk, Magdalena Sowa-Kucma, Agata Siwek, Urszula Doboszewska, Wojciech Piekoszewski, Piotr Gruca, Mariusz Papp, Gabriel Nowak; Antidepressant-like activity of magnesium in the chronic mild stress model in rats: alterations in the NMDA receptor subunits; 2014
- Fluoroquinolones-Associated Disability: It Is Not All in Your Head; Maya Freeman et al. 2021
- https://medcode.ch/ch/de/icds/ICD10-GM-2018/T3


